Benzyl chloride
- a-Chlorotoluene
- o-Chlorotoluene
- 1-Chloro-2-methyl benzene
- (Chloromethyl) benzene
- Chloromethylbenzene
- Tolyl chloride
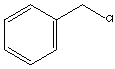
Benzyl chloride | |
| |
| Formula | C6H5CH2Cl |
| Structure |  |
| Description | Colorless liquid, unpleasant, irritating, pungent odor, lachrymator. |
| Uses | Manufacture benzyl compounds, perfumes, pharmaceutical products, dyes, synthetic tannins, & artificial resins. |
| Registry Numbers and Inventories. | |
| CAS | 100-44-7 |
| EC (EINECS/ELINCS) | 202-853-6 |
| EC Index Number | 602-037-00-3 |
| EC Class | Carcinogenic Category 2; Toxic; Harmful; Irritant |
| EC Risk Phrase | R 45 22 23 37/38 41 48/22 |
| EC Safety Phrase | S 53 45 |
| RTECS | XS8925000 |
| RTECS class | Tumorigen; Mutagen; Reproductive Effector |
| UN (DOT) | 1738 |
| Merck | 13,1131 |
| Beilstein/Gmelin | 471308 |
| Beilstein Reference | 4-05-00-00809 |
| RCRA | P028 |
| Swiss Giftliste 1 | G-1252 |
| Canada DSL/NDSL | DSL |
| US TSCA | Listed |
| Austrailia AICS | Listed |
| New Zealand | Listed |
| Japan ENCS (MITI) | Listed |
| Korea ECL | Listed |
| Properties. | |
| Formula | C7H7Cl |
| Formula mass | 126.59 |
| Melting point, °C | -41.2 |
| Boiling point, °C | 179.3 |
| Vapor pressure, mmHg | 1.6 (20 C) |
| Vapor density (air=1) | 4.36 |
| Saturation Concentration | 1316 ppm (0.13%) at 20 C (calculated) |
| Critical temperature | 411 |
| Critical pressure | 38.5 |
| Density | 1.103 g/cm3 (20 C) |
| Solubility in water | 0.465 g/L (30 C) |
| Viscosity | 1.28 cp (25 C) |
| Surface tension | 36.63 g/s2 (20 C) |
| Refractive index | 1.5415 (20 C) |
| Dipole moment | 1.87 D |
| Dielectric constant | 6.43 |
| Partition coefficient, pKow | 2.30 |
| Heat of vaporization | 46.2 kJ/mol |
| Heat of combustion | -3712 kJ/mol |
| Hazards and Protection. | |
| Storage | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a tightly closed container. Keep from contact with oxidizing materials. Store in a cool, dry, well-ventilated area away from incompatible substances. Store protected from moisture. |
| WHMIS | E B3 D1B D2A |
| Handling | Wash thoroughly after handling. Wash hands before eating. Remove contaminated clothing and wash before reuse. Use only in a well ventilated area. Do not get in eyes, on skin, or on clothing. Keep container tightly closed. Do not ingest or inhale. Do not allow contact with water. Discard contaminated shoes. Keep from contact with moist air and steam. |
| Protection | Eyes: Wear safety glasses and chemical goggles if splashing is possible. Skin: Wear appropriate protective gloves and clothing to prevent skin exposure. Clothing: Wear appropriate protective clothing to prevent skin exposure. |
| Respirators | A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant a respirator's use. Follow the OSHA respirator regulations found in 29CFR 1910.134 or European Standard EN 149. Always use a NIOSH or European Standard EN 149 approved respirator when necessary. |
| Small spills/leaks | Absorb spill with inert material, (e.g., dry sand or earth), then place into a chemical waste container. Clean up spills immediately, using the appropriate protective equipment. Use water spray to disperse the gas/vapor. Provide ventilation. Do not get water inside containers. |
| Disposal code | 2 |
| Stability | Unstable. Inhibitors such as propylene oxide, sodium carbonate solution, lime, or trimethylamine must be used to prevent polymerization. |
| Incompatibilities | Water, dimethyl sulfoxide, oxidizing material, steam. Corrodes all common metals except lead and nickel (explosive when heated). |
| Decomposition | Hydrogen chloride, chlorine, carbon monoxide, carbon dioxide. |
| Fire. | ||||
| Flash Point,°C | 52 | |||
| Autoignition, °C | 585 | |||
| Upper exp. limit, % | 7.1 | |||
| Lower exp. limit, % | 1.1 | |||
| Fire fighting | Wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. May accumulate static electrical charges, and may cause ignition of its own vapors. Forms explosive mixtures with air. Extinguishing media: Do NOT use water directly on fire. Use water spray to cool fire-exposed containers. Use foam, dry chemical, or carbon dioxide. Do NOT get water inside containers. | |||
| Fire potential | Combustible liquid and vapor. | |||
| Hazards | It burns but does not ignite readily. It may ignite combustibles. When heated to decomposition, it emits toxic and corrosive fumes. Some organic chlorides decompose to yield phosgene. Incompatible with active metals such as copper, aluminum, magnesium, iron, zinc, and tin and keep from strong oxidizing agents. Avoid contact with acids or acid fumes. Keep separate from oxidizing materials. May become unstable at elevated temperatures and pressures; may react with water resulting in some nonviolent release of energy. Polymerizes with evolution of heat and hydrogen chloride when in contact with all common metals except nickel and lead. | |||
| Combustion products | Fire will produce irritating, corrosive and/or toxic gases. | |||
| NFPA | Health | 3 | ||
| Flammability | 2 | |||
| Reactivity | 1 | |||
| Health. | |
| Exposure limit(s) | TLV: 1 ppm; 5.2 mg/m3 (as TWA) (ACGIH 1992-1993). OSHA PEL: TWA 1 ppm (5 mg/m3) NIOSH REL: C 1 ppm (5 mg/m3) 15-minute NIOSH IDLH: 10 ppm |
| Carcinogin | G-A3, I-2A, CP65 |
| Poison_Class | 3 |
| Exposure effects | Mild leukopenia(abnormally low number of circulating white blood cells), liver function disturbances and kidney problems. |
| Ingestion | Corrosive. Swallowing can cause severe burns of the mouth, throat, and stomach, leading to death. Can cause sore throat, vomiting, diarrhea. May also cause systemic poisoning with symptoms paralleling inhalation. |
| Inhalation | Causes severe irritation of upper respiratory tract with coughing, burns, breathing difficulty, and possible coma. Causes chemical burns to the respiratory tract. Inhalation may be fatal as a result of spasm, inflammation, edema of the larynx and bronchi, chemical pneumonitis and pulmonary edema. |
| Skin | Prolonged and/or repeated contact may cause irritation and/or dermatitis. Contact with liquid is corrosive and causes severe burns and ulceration. |
| Eyes | Contact with liquid or vapor causes severe burns and possible irreversible eye damage. Lachrymator. |
First aid |
|
| Ingestion | Never give anything by mouth to an unconscious person. Get medical aid immediately. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water. |
| Inhalation | Get medical aid immediately. Remove from exposure to fresh air immediately. If breathing is difficult, give oxygen. DO NOT use mouth-to-mouth respiration. If breathing has ceased apply artificial respiration using oxygen and a suitable mechanical device such as a bag and a mask. |
| Skin | Get medical aid immediately. Flush skin with plenty of soap and water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse. Destroy contaminated shoes. |
| Eyes | Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately. Do NOT allow victim to rub or keep eyes closed. Extensive irrigation is required (at least 30 minutes). |
| Transport. | ||
| UN number | 1738 |  |
| Response guide | 156 | |
| Hazard class | 6.1 | |
| Packing Group | II | |
| USCG CHRIS Code | BCL | |
| USCG Compatatibility Group | 36 Halogenated hydrocarbons | |
| HS Code | 2903 69 90 | |
| Std. Transport # | 4921209 | |
| IMO Pollution Category | B | |
| IMO Hazard code | S/P | |